
Science
By Josh Paigen
·Published
·Last Updated
·8 min read

Men don't have a menopause equivalent, and that's led to a persistent myth: that male fertility doesn't decline with age, but it does. The decline is gradual rather than sudden, but it's real, measurable, and starts earlier than most men expect.
Male fertility begins declining gradually from the mid-30s onward. Motility declines about 0.7% per year, DNA fragmentation increases steadily, semen volume decreases continuously, and testosterone drops roughly 1% annually after age 30.1,2 A meta-analysis of 90 studies covering 93,839 men confirmed consistent age-dependent declines across most major semen parameters.2
Here's what I want to be clear about: this is not a reason to panic. It's a reason to understand the biology and act on what's modifiable. Because while you can't stop aging, you can control the lifestyle factors that either accelerate or slow these changes.
Female fertility has a well-defined biological endpoint. Menopause marks the cessation of ovulation, creating a clear window that closes.
Male fertility doesn't work that way. There's no equivalent shutdown. Men can and do father children into their 60s, 70s, and beyond.
But "technically possible" is not the same as "optimal." Kidd and colleagues' comprehensive review established that semen quality deteriorates progressively with advancing paternal age, with measurable changes in volume, motility, and morphology beginning in the late 30s and accelerating after 45.1 The decline doesn't have a cliff edge. It's more like a slope that gets steeper the further you go.
The reason this matters is that most men don't think about it until they're already on the slope. Average paternal age has been climbing steadily, with more men fathering children in their late 30s and 40s than in previous generations. Understanding where you sit on the curve gives you time to act.
Johnson and colleagues' meta-analysis of 90 studies (93,839 subjects) is the most comprehensive dataset on age-related semen changes.2 Combined with Kidd et al. and Hellstrom et al.'s data on men over 45, here's how the major parameters shift across decades.1,6
| Parameter | 20s | 30s | 40s | 50s+ |
|---|---|---|---|---|
| Sperm Concentration million/mL | 70–90 | 60–80 | 45–70 | 35–60 |
| Progressive Motility % swimming forward | 55–65% | 48–58% | 40–52% | 35–45% |
| Normal Morphology % strict criteria | 8–12% | 6–10% | 4–8% | 3–6% |
| Semen Volume mL per ejaculate | 3.0–4.0 | 2.5–3.5 | 2.0–3.0 | 1.5–2.5 |
| DNA Fragmentation DFI % | 10–15% | 15–20% | 20–30% | 25–40% |
| Testosterone ng/dL (approximate) | 600–750 | 500–700 | 400–600 | 350–550 |
Ranges are approximate population averages derived from Kidd et al. (2001),1 Johnson et al. (2015),2 and Hellstrom et al. (2006).6 Individual variation is significant. Global sperm counts have declined substantially over the past several decades,13 which means real-world averages today may be lower than what these studies reported.
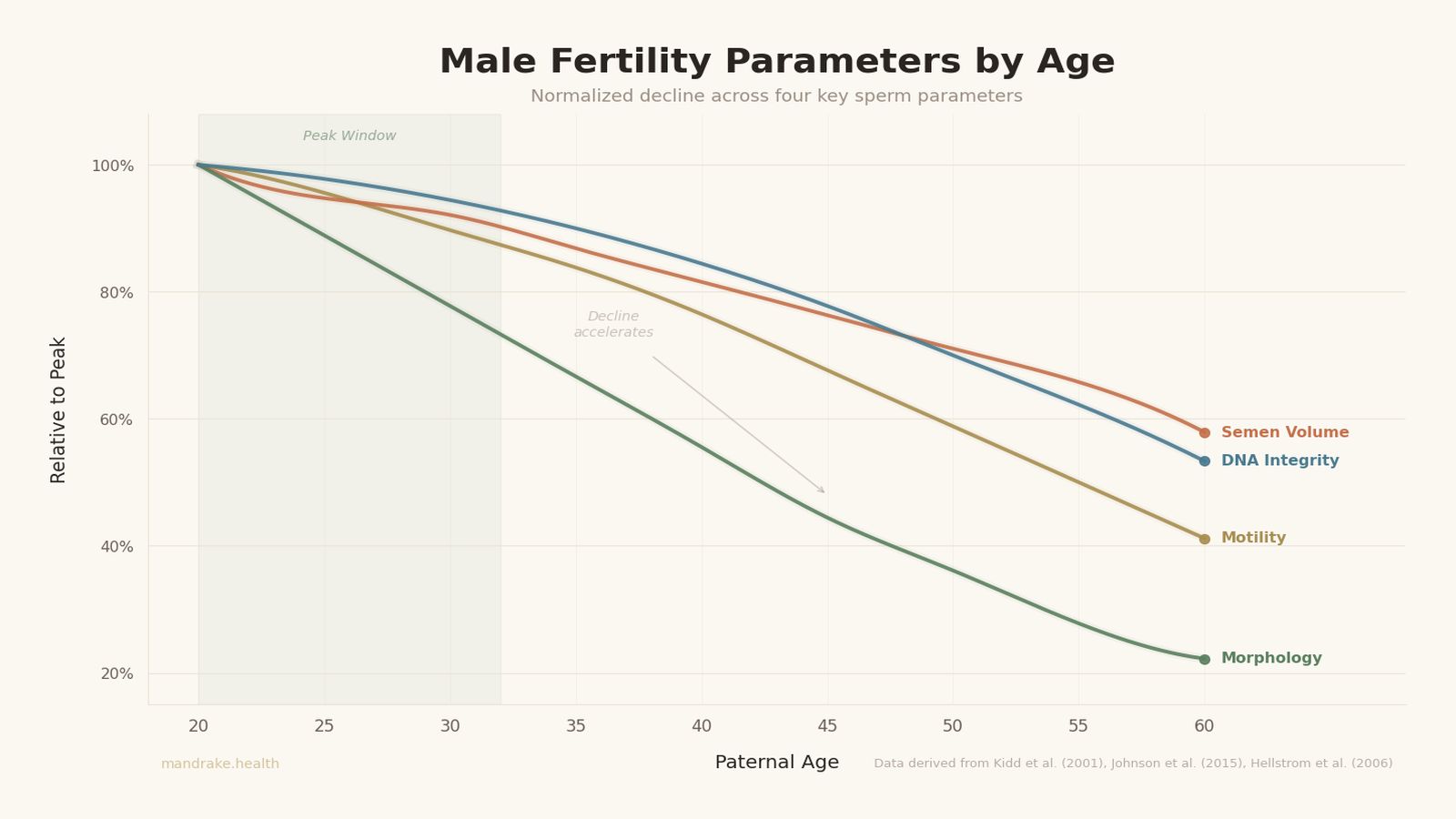
Sperm concentration peaks in the mid-20s to early 30s at roughly 70 to 90 million per milliliter. Cross-sectional studies report gradual declines with age,16 though Johnson and colleagues' meta-analysis noted that the decline in concentration was not statistically significant across all pooled data.2 Individual variation is substantial: some men maintain high counts into their 60s, while others show measurable decline by their late 30s.
Motility (the percentage of sperm that swim effectively) declines approximately 0.7% per year starting as early as the mid-20s.2 By the 50s, progressive motility may be 15 to 25% below what it was at 25. This is one of the earliest parameters to show age effects.
Morphology (the percentage of normally shaped sperm) also worsens with age. The WHO defines normal morphology as 4% or greater using strict criteria. Aging shifts the distribution, increasing the proportion of abnormally shaped sperm.
Semen volume decreases with age, reflecting reduced function of the seminal vesicles and prostate. Kidd and colleagues found a continuous decline of roughly 0.03 mL per year from the early 20s through the 80s, with no specific threshold age.1 The decline is modest but measurable over decades.
DNA fragmentation increases with age, driven by cumulative oxidative stress and declining repair efficiency. Men over 40 typically have higher DFI values than men in their 20s, even when other parameters appear adequate.3
Testosterone declines approximately 1% per year after age 30 to 35, reflecting gradual Leydig cell decline.8 By age 50, testosterone levels are often 15 to 20% below peak. This hormonal shift affects spermatogenesis directly, since testosterone is the primary driver of sperm production.
The numbers above translate into real reproductive consequences. Not guarantees of failure, but measurable shifts in probability.
Time to pregnancy increases. Sartorius and Nieschlag's review found that men over 40 face longer time to conception, independent of female partner age.3 When both partners are older, the effect compounds.
Miscarriage risk rises. Higher DNA fragmentation rates in older men are associated with increased risk of early pregnancy loss. This is the same mechanism covered in the DNA fragmentation post: damaged DNA can initiate fertilization but the resulting embryo may not be viable.3
De novo mutation rate increases. De novo mutations are brand-new genetic errors that weren't present in either parent's own DNA. They arise during the cell divisions that produce sperm, and they get passed directly to the child. Kong and colleagues' landmark 2012 study in Nature found that the number of these new mutations in offspring increases by roughly two additional mutations for every year of paternal age.4
To put that in perspective: a 20-year-old father passes on roughly 25 de novo mutations. A 40-year-old passes on roughly 65. Most of these mutations are harmless, but the more you accumulate, the higher the statistical probability that one lands in a gene that matters. Research has linked higher de novo mutation counts to increased risk of certain neurodevelopmental conditions in offspring.
Crow's earlier work established why this happens on the paternal side specifically: sperm-producing cells divide continuously throughout a man's life, and each division is another opportunity for a copying error. Eggs, by contrast, are formed before birth and don't undergo the same ongoing replication.5
ART outcomes are affected. A meta-analysis of over 32,000 IVF cycles found that paternal age under 40 was associated with 1.65 to 2.10 times higher pregnancy and live birth rates compared to advanced paternal age in cycles using the couple's own eggs.9
None of this means conception is impossible at older ages. It means the margin for error narrows. And it means the lifestyle factors that influence sperm quality become more important, not less, as the years progress.
The parameters above measure the physical characteristics of sperm. But age also changes something less visible: the epigenome.
Sperm carry epigenetic information through DNA methylation patterns, which influence how genes are expressed in offspring without changing the DNA sequence itself. Research has shown that aging alters these methylation patterns, with the vast majority of affected regions (139 of 147 identified) becoming hypomethylated (less methylated) and a smaller subset becoming hypermethylated.10 The affected regions concentrate in developmental and metabolic gene networks.
Jenkins and Carrell's work demonstrated that paternal aging affects not just DNA methylation but also histone modifications and non-coding RNA profiles in sperm, all of which can influence offspring development through neurodevelopmental and metabolic pathways.11 Interestingly, sperm telomere length tends to increase with age (unlike most somatic cells), but the functional implications of this remain an active area of research.
This is where the conversation shifts from "can I conceive?" to "what am I passing on?" The epigenetic changes associated with aging are influenced by the same lifestyle factors that affect sperm parameters: oxidative stress, nutrition, toxin exposure, sleep quality, and chronic inflammation. The five S.P.E.R.M. pillars protect the epigenetic information your sperm carry forward, not just the count and motility numbers.
Every study cited in this post measures decline by chronological age, the number of years since you were born. That's the only variable researchers can easily standardize across large populations. But chronological age is a rough proxy for what's actually happening at the cellular level, and two men who are both 42 can have very different biological realities.
Biological age refers to the functional state of your cells, tissues, and organ systems, shaped by accumulated damage, repair capacity, metabolic health, and mitochondrial function. Researchers now measure it using epigenetic clocks, which track DNA methylation patterns that change predictably with aging. Pilsner and colleagues developed a sperm-specific epigenetic clock and found that men with higher sperm epigenetic age had a 17% lower cumulative probability of pregnancy after 12 months, independent of their chronological age.14 Notably, current smokers in the same study showed accelerated sperm epigenetic aging, which demonstrates that lifestyle directly shifts these biological age markers.
This distinction matters because it means your chronological age is not a verdict. A 44-year-old who has spent the past two years optimizing his metabolic health, reducing oxidative stress, sleeping well, and avoiding toxin exposure may have a meaningfully younger biological profile than a 36-year-old who smokes, drinks heavily, and sleeps five hours a night. The decline curves in the chart above represent population averages, and your individual position on those curves depends heavily on how you've been living.
A comprehensive review in Human Reproduction Update confirmed that age-dependent epigenetic changes in sperm are not fixed or uniform across individuals, and that lifestyle and environmental exposures influence the rate at which these changes accumulate.15 This is the core argument for preconception optimization: you can't change the year you were born, but you can change the biological environment your sperm develop in.
The data above is not a death sentence, it's a starting point for action.
Because spermatogenesis is continuous and takes 72 to 74 days per cycle, the sperm your body produces are always a reflection of your current conditions.12 Age sets the baseline, and lifestyle determines how far above or below that baseline you sit.
Antioxidant support becomes more important with age because cumulative oxidative stress is the primary driver of age-related sperm damage. The same nutrients covered in the supplements post (CoQ10, vitamin C, vitamin E, selenium, zinc) directly counter this mechanism.
Toxin avoidance matters more with age because your body's detoxification capacity declines. Reducing exposure to endocrine disruptors, avoiding smoking and excess alcohol, and minimizing heat exposure protect an increasingly vulnerable system.
DNA fragmentation testing gives you actionable data. If you're over 40 and planning conception, knowing your DFI baseline lets you track whether your interventions are working. Having real numbers to track makes the process significantly easier to stick with.
For me, this data isn't abstract. My wife and I conceived our daughter when I was 36, after ten months of dialing in our nutrition, toxin exposure, sleep, and stress. We conceived our son when I was 39. Both times, the preparation mattered more because of where I sat on the age curve, not less.
The thing is, most men over 35 don't think about preconception optimization at all. I would advocate for treating it with the same seriousness women do, not because male fertility is as time-sensitive as female fertility, but because the gradual nature of male decline makes it easy to ignore until it becomes a problem.
The three-month spermatogenic window is your ally at any age, and the sperm you produce three months from now will reflect whatever you decide to do today.
If you're over 35 and thinking about conception, or if you've seen age-related changes in your lab work and want a plan, that's exactly what the Mandrake Method addresses. We build a personalized protocol around your specific numbers, your age, and your timeline, calibrated to where you actually are right now rather than where a generic guide assumes you might be.
Sperm parameters begin gradually declining around age 35 to 40. Some cross-sectional studies report concentration declines with age, though the evidence is mixed across meta-analyses. Motility declines about 0.7% per year, and DNA fragmentation increases.1,2
The decline accelerates after age 45. Unlike female fertility, there's no sharp cutoff, but the gradual change is real and measurable across every major semen parameter.
Sperm concentration is generally highest in men aged 25 to 35 (approximately 70 to 90 million per milliliter on average). Cross-sectional studies report gradual declines with age,1,6 though large meta-analyses show mixed results on whether concentration specifically declines.2 Individual variation is significant, and lifestyle factors can accelerate or slow age-related changes substantially.
Yes, but the risks increase with age. Men over 45 have higher rates of de novo genetic mutations in sperm (approximately 65 mutations at age 40 versus 25 at age 20), increased DNA fragmentation, and longer time to pregnancy.4,3 Lifestyle optimization becomes more important with advancing age to mitigate these effects.
Yes. Motility declines approximately 0.7% per year. Morphology worsens. DNA fragmentation increases. Semen volume decreases. Testosterone drops roughly 1% per year after age 30.1,2,8
These changes are gradual but cumulative. The good news: many age-related changes are amplified by poor lifestyle habits, meaning optimization still makes a measurable difference.
The same S.P.E.R.M. framework applies with extra emphasis on antioxidant support (CoQ10, vitamins C and E, selenium, zinc), toxin avoidance, and DNA fragmentation testing. Men over 40 benefit disproportionately from lifestyle optimization because they're starting from a lower baseline. Every intervention has a higher relative impact when the margin for error is narrower.
1 Kidd SA, Eskenazi B, Wyrobek AJ. Effects of male age on semen quality and fertility: a review of the literature. Fertil Steril. 2001;75(2):237-248. doi:10.1016/S0015-0282(00)01679-4
2 Johnson SL, Dunleavy J, Gemmell NJ, Nakagawa S. Consistent age-dependent declines in human semen quality: a systematic review and meta-analysis. Ageing Res Rev. 2015;19:22-33. doi:10.1016/j.arr.2014.10.007
3 Sartorius GA, Nieschlag E. Paternal age and reproduction. Hum Reprod Update. 2010;16(1):65-79. doi:10.1093/humupd/dmp027
4 Kong A, Frigge ML, Masson G, et al. Rate of de novo mutations and the importance of father's age to disease risk. Nature. 2012;488(7412):471-475. doi:10.1038/nature11396
5 Crow JF. The origins, patterns and implications of human spontaneous mutation. Nat Rev Genet. 2000;1(1):40-47. doi:10.1038/35049558
6 Hellstrom WJ, et al. Semen and sperm reference ranges for men 45 years of age and older. J Androl. 2006;27(3):421-428. doi:10.2164/jandrol.05156
7 Kovac JR, Addai J, Smith RP, Coward RM, Lamb DJ, Lipshultz LI. The effects of advanced paternal age on fertility. Asian J Androl. 2013;15(6):723-728. doi:10.1038/aja.2013.92
8 Tsujimura A. The relationship between testosterone deficiency and men's health. World J Mens Health. 2013;31(2):126-135. doi:10.5534/wjmh.2013.31.2.126
9 Murugesu S, Kasaven LS, Petrie A, et al. Does advanced paternal age affect outcomes following assisted reproductive technology? A systematic review and meta-analysis. Reprod Biomed Online. 2022;45(2):283-331. doi:10.1016/j.rbmo.2022.03.031
10 Jenkins TG, Aston KI, Pflueger C, Cairns BR, Carrell DT. Age-associated sperm DNA methylation alterations: possible implications in offspring disease susceptibility. PLoS Genet. 2014;10(7):e1004458. doi:10.1371/journal.pgen.1004458
11 Jenkins TG, Carrell DT. The sperm epigenome and potential implications for the developing embryo. Reproduction. 2012;143(6):727-734. doi:10.1530/REP-11-0450
12 Amann RP. The cycle of the seminiferous epithelium in humans: a need to revisit? J Androl. 2008;29(5):469-487. doi:10.2164/jandrol.107.004655
13 Levine H, Jørgensen N, Martino-Andrade A, et al. Temporal trends in sperm count: a systematic review and meta-regression analysis of samples collected globally in the 20th and 21st centuries. Hum Reprod Update. 2023;29(2):157-176. doi:10.1093/humupd/dmad006
14 Pilsner JR, Saddiki H, Whitcomb BW, et al. Sperm epigenetic clock associates with pregnancy outcomes in the general population. Hum Reprod. 2022;37(7):1581-1591. doi:10.1093/humrep/deac084
15 Ashapkin VV, Suvorov A, Pilsner JR, Krawetz SA, Sergeyev O. Age-associated epigenetic changes in mammalian sperm: implications for offspring health and development. Hum Reprod Update. 2023;29(1):24-66. doi:10.1093/humupd/dmac033